WHO WE ARE Intra-ImmuSG Pte Ltd (IISG) is a Phase 2 clinical-stage biotechnology company
At IISG, we are developing new cancer drugs to specifically target tumors, and with minimal side effects for patients.
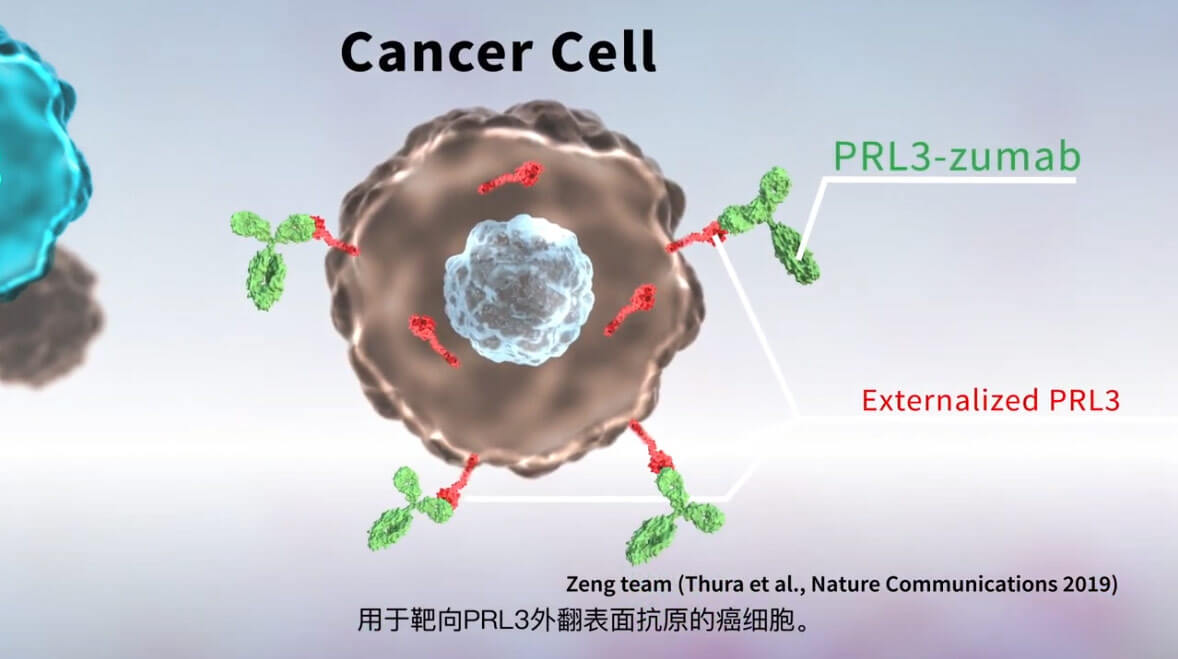
PRL3 is an intracellular tumor-specific antigen overexpressed in about 80.6% of cancer patients. Conventionally, chemotherapy is used to target molecules that reside in cancer cells, such as PRL3 intracellular oncoprotein. Antibody drugs are not used to target intracellular oncotargets due to their inability to cross cell membranes. However, we discovered that PRL3 intracellular oncotarget could be externalised as an ‘extracellular’ antigen on the surface of cancer cells and to be targeted by antibodies to trigger the host’s immune system via ADCC/ADCP (Thura et al., Nature Communications 2019). This discovery opens a new avenue for cancer immunotherapy against a spectrum of intracellular oncotargets.
PRL3-zumab is our first Flagship antibody drug and is representative of an innovative and disruptive approach in a new era of cancer immunotherapy. We anticipate that PRL3-zumab will bring clinical benefits to majority of PRL3 positive cancer patients.
ABOUT US
For Patients
IISG is established with the aim to develop more effective cancer immunotherapies with minimal side effects as compared to traditional cancer drugs.
We are committed to improve the lives of cancer patients across the world. Through the development of our high affinity monoclonal antibodies, we strive to provide patients with more effective, safer and long-lasting treatment options for a broad range of cancers.
READ MORE